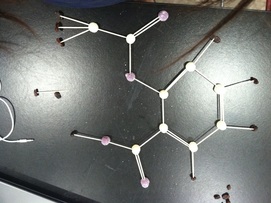
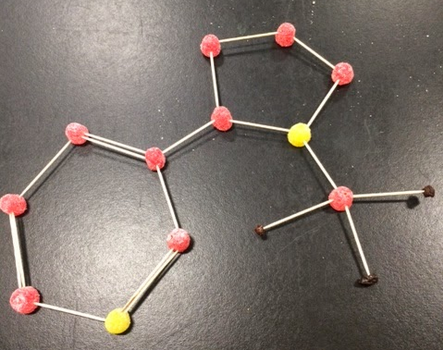
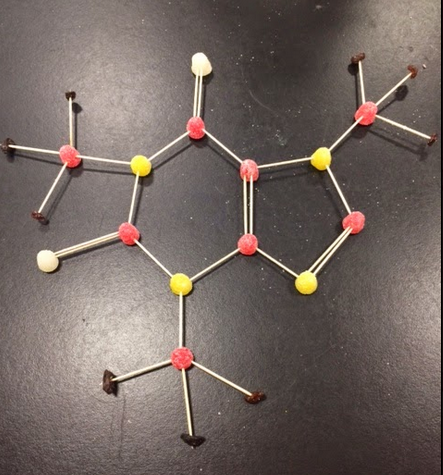
In this lab we constructed molecule structures. We used Tooth picks as bonds, raisins as hydrogen atoms, and gumdrops as all the other atoms. We were given a sheet with compounds with both inorganic molecules and organic molecules and we had to create what we though the right structure was. These are the ones that my group and I made.
INORGANIC MOLECULE STRUCTURES
| | SCI6 |

S2H
ORGANIC MOLECULE STRUCTURES
This activity that we did was a difficult at the beginning because we where just trying to put them together without considering the electrons. However when we started working on figuring out how many electrons each atom had it got easier. We learned that each atom wants to be as stable as possible so the bigger atoms take other atoms (like hydrogen) to get 8 electrons.








 RSS Feed
RSS Feed
